A new incentive structure under the IRA
Ms. Valder Curran explained that the IRA includes major provisions relevant for drug pricing and reimbursement, including an entirely new Drug Price Negotiation Program, which seeks to lower the prices of certain Medicare high spend drugs by imposing a maximum fair price (MFP) on specified qualifying single source drugs (QSSD). Under the program, certain New Drug Application (NDA) drugs at least seven years post-approval and certain Biologics License Application (BLA) biologics at least eleven years post-approval and for which there is no generic / biosimilar on the market are subject to “negotiation” and application of an MFP. Our team has previously outlined key details of these provisions here and here.
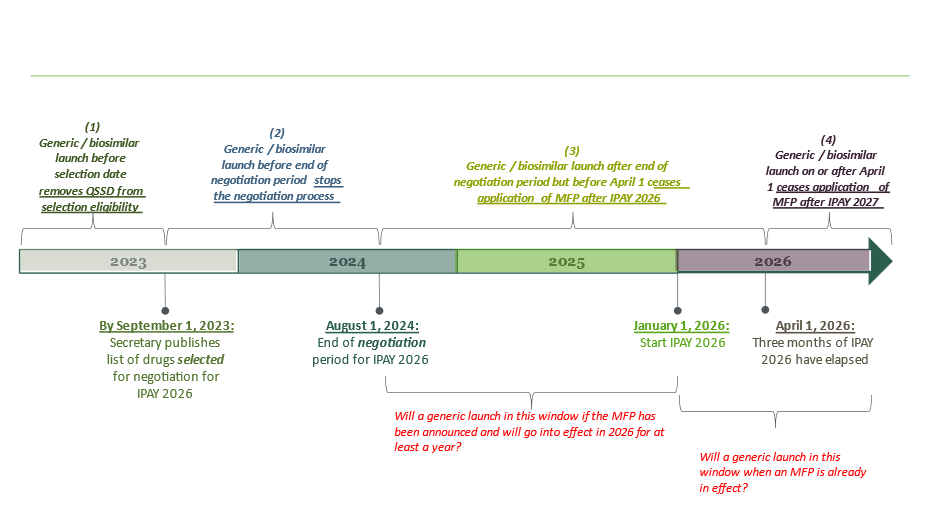
She explained the relevance of various IRA provisions for this IP-focused audience in framing the litigation and licensing considerations that the panelists presented in more detail. She also discussed the importance of timing, pointing out that a generic / biosimilar must be marketed before the end of the “negotiation” period in order to avoid application of MFP, with later entry creating other unique dis/incentives for generic entrants and their branded counterparts. Ms. Valder Curran discussed the factors underlying the timeline depicted below, which shows the selection and negotiation dates for initial price applicability year (IPAY) 2026, and highlights the impact of a generic / biosimilar launch, respectively:
- before the QSSD selection date;
- before the end of the negotiation period;
- after the end of the negotiation period but through the first three months of IPAY 2026, or
- on or after April 1 of IPAY.
 Ms. Valder Curran and Mr. Hadad also highlighted a number of examples of how the IRA changes the historical incentive structure for investment in product research and development in the broader context of patient centered innovation.
Ms. Valder Curran and Mr. Hadad also highlighted a number of examples of how the IRA changes the historical incentive structure for investment in product research and development in the broader context of patient centered innovation.
Patent litigation strategies: balancing the patent cliff with the new “MFP cliff”
Shifting to litigation strategies, Ms. Gundt noted that companies will still need patents in order to protect their products and to litigate. However, she noted, “patent and regulatory exclusivities will not protect from an MFP.”
She then discussed how companies should assess the impact of the IRA’s new “MFP cliff” as compared to the well-known “patent cliff” when evaluating possible litigation scenarios, offering a number of factors (including timelines for protection, revenue impact of price controls vs. generic / biosimilar entry, protecting alternative compounds vs. multiple uses) that companies should evaluate for each of their product(s) / portfolio(s). Ms. Gundt then detailed several revenue impact scenarios and discussed the impact of the generic and biosimilar launch timing considerations framed by Ms. Valder Curran. She discussed, for example, that there may be scenarios where the appropriate facilitation of a generic entrant may be more attractive than imposition of an MFP. She also noted that stakeholders should be on the alert for the upcoming first round of MFPs to glean additional insights to inform their ongoing analyses. Ms. Gundt also noted that settlements and / or agreements may ultimately take a larger role in informing litigation scenario planning, but cautioned that companies will also need to consider the implication of possible antitrust issues in any analysis.
Licensing strategies: taking a health check
In the context of ongoing and future collaboration arrangements, Mr. Taylor noted that there are number of IRA-related considerations to consider, for both licensors and licensees. He outlined a number of new risk considerations from the perspective of each side of such transactions, as well as possible preferences that each side might consider advocating for in order to maintain control / flexibility in their agreements. For example, post-IRA, licensees may have higher interest in retaining rights to backup compounds and combination therapies, as well as increased need for flexibility in sublicensing and launch strategies, and a desire to ensure the diligence and economic provisions account for the impact of the MFP. Licensors will be focused on ensuring they are appropriately compensated for granting rights to backups and combination therapies, as well as ensuring a licensee carries out a timely launch. Our team has also described the impacts of the IRA on licenses and collaborations here.
Mr. Taylor also presented a framework that companies could use in evaluating their existing and future product(s) / portfolio(s) for MFP risk, based on, for example, the projected initial IPAY for a product in a particular indication and the anticipated timing of any generic / biosimilar entry. He also explored whether (partial) out-licensing opportunities might provide opportunities to mitigate these risks across molecule(s), especially where a therapy may offer patient benefits across multiple possible indication(s).
Next steps
As the IRA’s impact on product development and commercialization strategies becomes more clear, manufacturers should ensure they are also considering how the IRA will impact their patent prosecution and litigation strategies, as well as the terms of any collaboration or other partnering arrangement where IP rights are shared or transferred. Every scenario will be subject to its own unique considerations, but certain foundational principles and considerations can help any manufacturer better consider how the IRA will affect its IP portfolio. Our panel provided frameworks to help industry stakeholders do just that.
The full presentation materials are available here. If you would like to request a copy of the recording, please contact our team.
Please contact the authors or the Hogan Lovells attorneys with whom you regularly work for guidance on your organization’s specific launch needs.
Authored by Alice Valder Curran, Miriam Gundt, and Cullen Taylor.

